Development of a Mild and Robust Method for Palladium Catalyzed Cyanation on Large Scale | SpringerLink

Pd/BIPHEPHOS is an Efficient Catalyst for the Pd‐Catalyzed S‐Allylation of Thiols with High n‐Selectivity - Schlatzer - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library
Labeling and Natural Post-Translational Modification of Peptides and Proteins via Chemoselective Pd-Catalyzed Prenylation of Cys

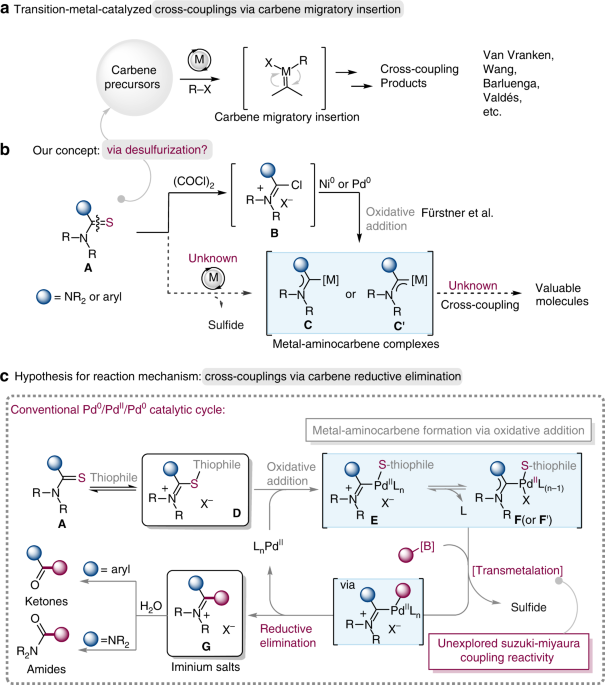
Palladium-catalyzed carbon-sulfur or carbon-phosphorus bond metathesis by reversible arylation | Science
Tackling poison and leach: catalysis by dangling thiol–palladium functions within a porous metal–organic solid - Chemical Communications (RSC Publishing) DOI:10.1039/C5CC00140D
![PDF] Tackling poison and leach: catalysis by dangling thiol-palladium functions within a porous metal-organic solid. | Semantic Scholar PDF] Tackling poison and leach: catalysis by dangling thiol-palladium functions within a porous metal-organic solid. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/eba13f8c85b1ac6daf9c2c9fdb588e83c7326980/3-Figure2-1.png)
PDF] Tackling poison and leach: catalysis by dangling thiol-palladium functions within a porous metal-organic solid. | Semantic Scholar

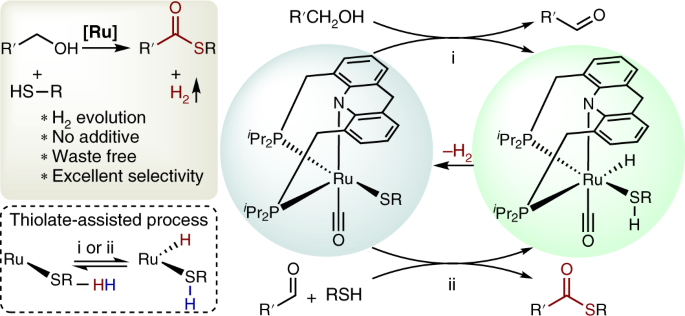
Thiol Treatment Creates Selective Palladium Catalysts for Semihydrogenation of Internal Alkynes - ScienceDirect
![PDF] Tackling poison and leach: catalysis by dangling thiol-palladium functions within a porous metal-organic solid. | Semantic Scholar PDF] Tackling poison and leach: catalysis by dangling thiol-palladium functions within a porous metal-organic solid. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/eba13f8c85b1ac6daf9c2c9fdb588e83c7326980/4-Figure3-1.png)
PDF] Tackling poison and leach: catalysis by dangling thiol-palladium functions within a porous metal-organic solid. | Semantic Scholar
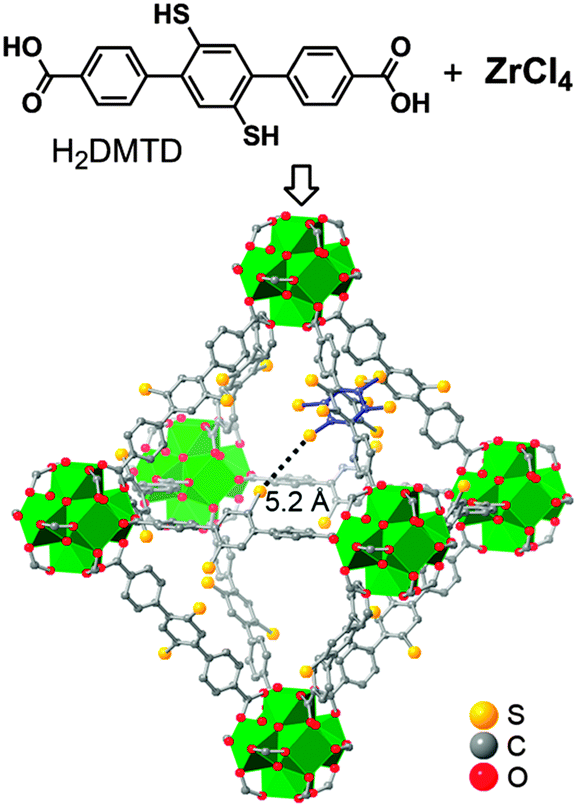
Tackling poison and leach: catalysis by dangling thiol–palladium functions within a porous metal–organic solid - Chemical Communications (RSC Publishing) DOI:10.1039/C5CC00140D

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Influence of selective catalyst poisons on the reaction progress of... | Download Scientific Diagram

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Palladium‐Catalyzed C–S Bond Formation of Stable Enamines with Arene/Alkanethiols: Highly Regioselective Synthesis of β‐Amino Sulfides - Jiang - 2016 - European Journal of Organic Chemistry - Wiley Online Library

Palladium-catalyzed carbon-sulfur or carbon-phosphorus bond metathesis by reversible arylation | Science

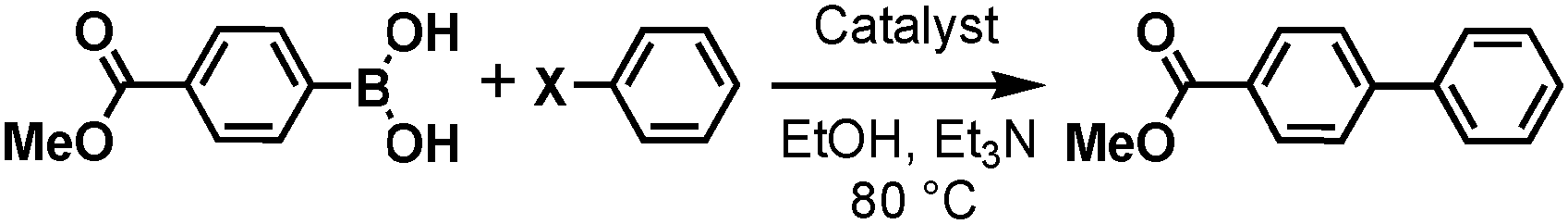
Thiol-Functionalized Ethylene Periodic Mesoporous Organosilica as an Efficient Scavenger for Palladium: Confirming the Homogeneous Character of the Suzuki Reaction. - Abstract - Europe PMC
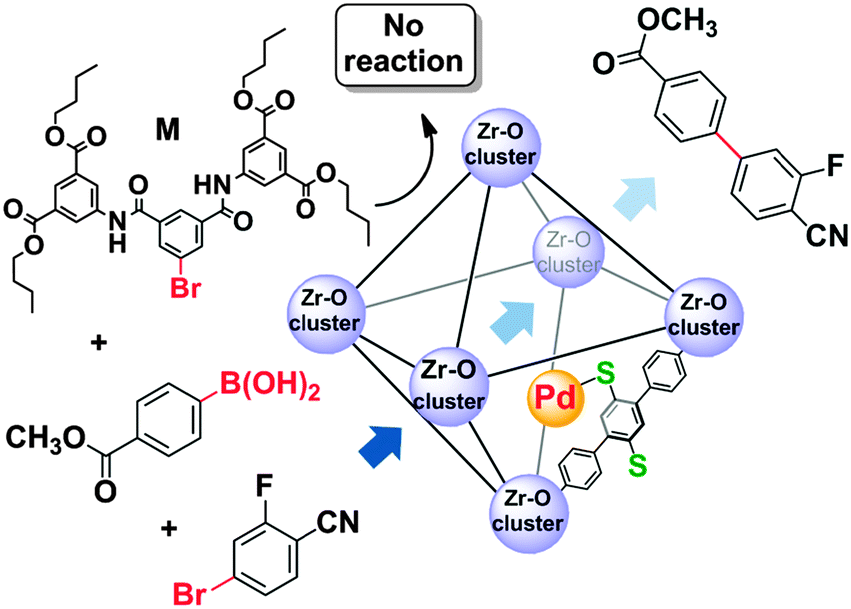
Tackling poison and leach: catalysis by dangling thiol–palladium functions within a porous metal–organic solid - Chemical Communications (RSC Publishing) DOI:10.1039/C5CC00140D