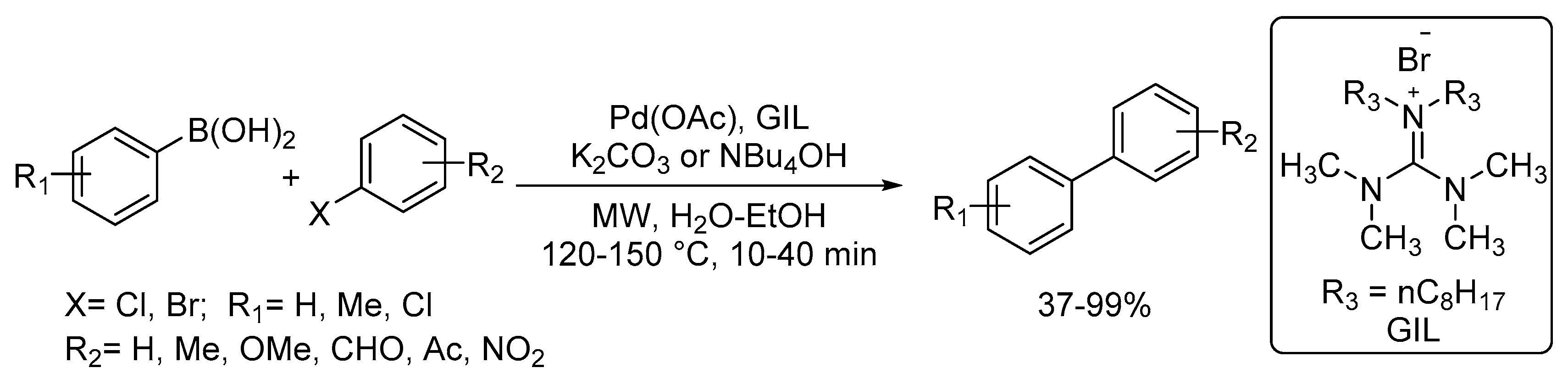
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML

Studies on Pd/NiFe 2 O 4 catalyzed ligand-free Suzuki reaction in aqueous phase: synthesis of biaryls, terphenyls and polyaryls – topic of research paper in Chemical sciences. Download scholarly article PDF and
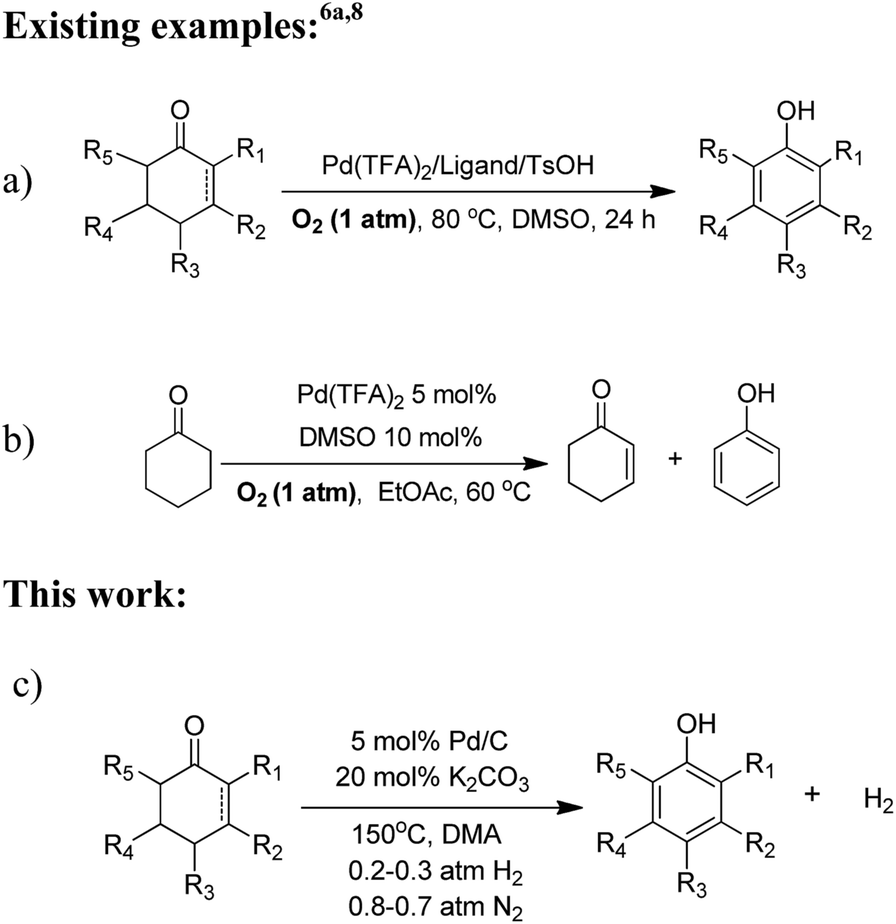
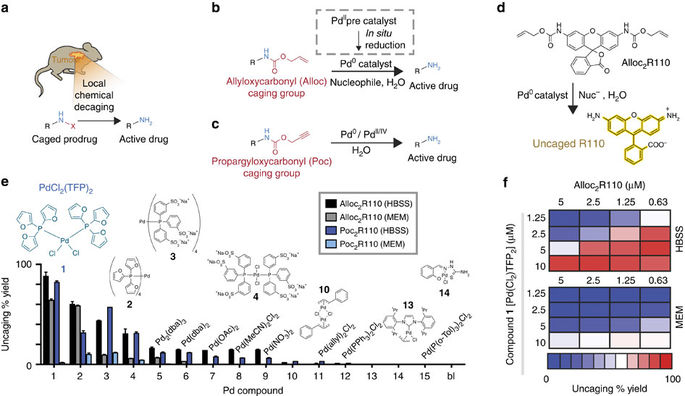
Reaction-activated palladium catalyst for dehydrogenation of substituted cyclohexanones to phenols and H2 without oxidants and hydrogen acceptors - Chemical Science (RSC Publishing)

Synthesis and Characterization of Palladium Supported Amino Functionalized Magnetic-MOF-MIL-101 as an Efficient and Recoverable Catalyst for Mizoroki–Heck Cross-Coupling | SpringerLink

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Supported Palladium Catalysts In Organic Synthesis: Kumbhar, Arjun, Salunkhe, Rajashri: 9783659449758: Amazon.com: Books

Synthesis of Biaryls via Decarbonylative Palladium-Catalyzed Suzuki-Miyaura Cross-Coupling of Carboxylic Acids - ScienceDirect

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds
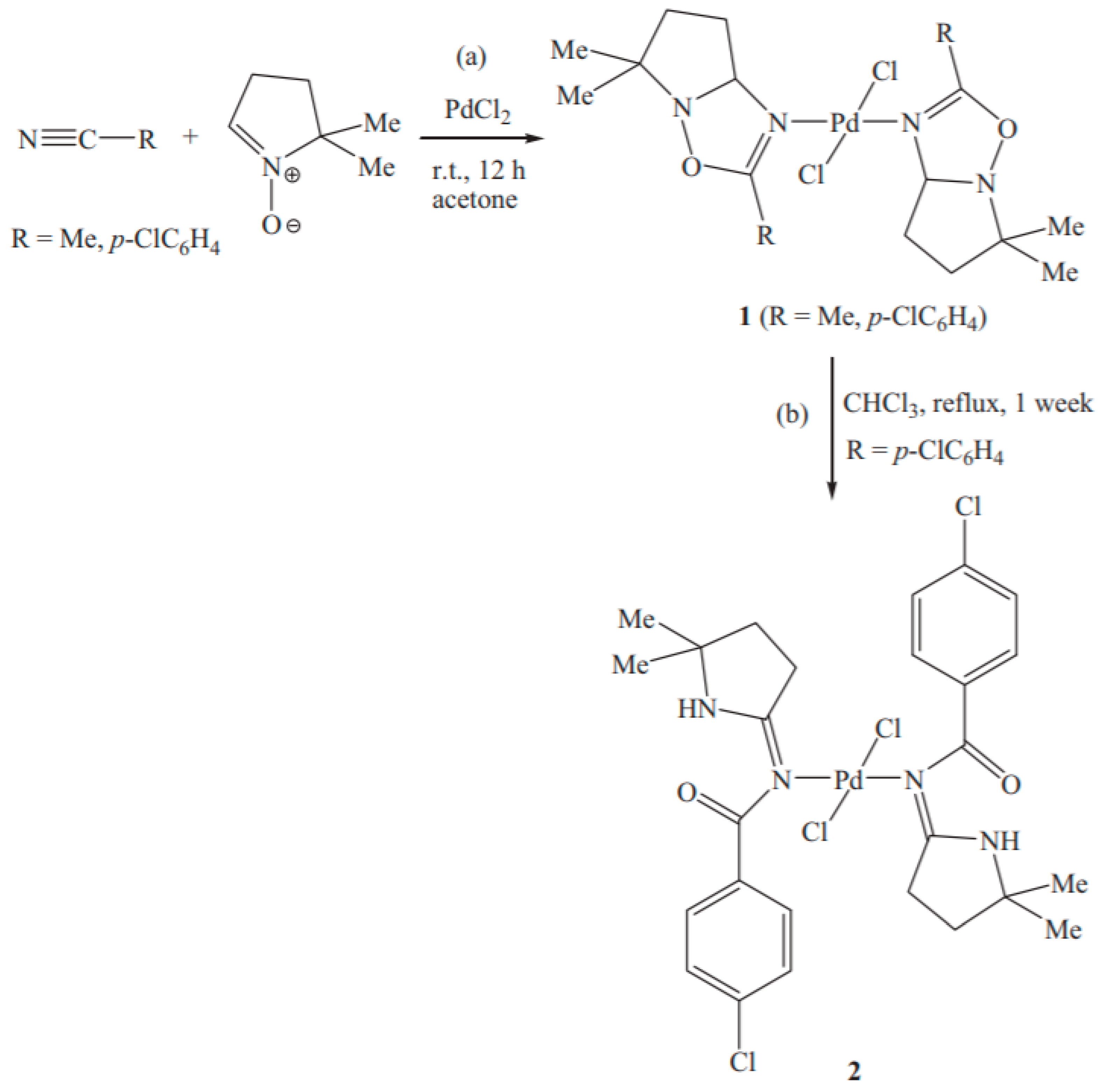
Catalysts | Free Full-Text | Recent Developments in the Immobilization of Palladium Complexes on Renewable Polysaccharides for Suzuki–Miyaura Cross-Coupling of Halobenzenes and Phenylboronic Acids | HTML

Catalysts | Free Full-Text | Recent Developments in the Immobilization of Palladium Complexes on Renewable Polysaccharides for Suzuki–Miyaura Cross-Coupling of Halobenzenes and Phenylboronic Acids | HTML

Factorial design evaluation of the Suzuki cross-coupling reaction using a magnetically recoverable palladium catalyst - ScienceDirect
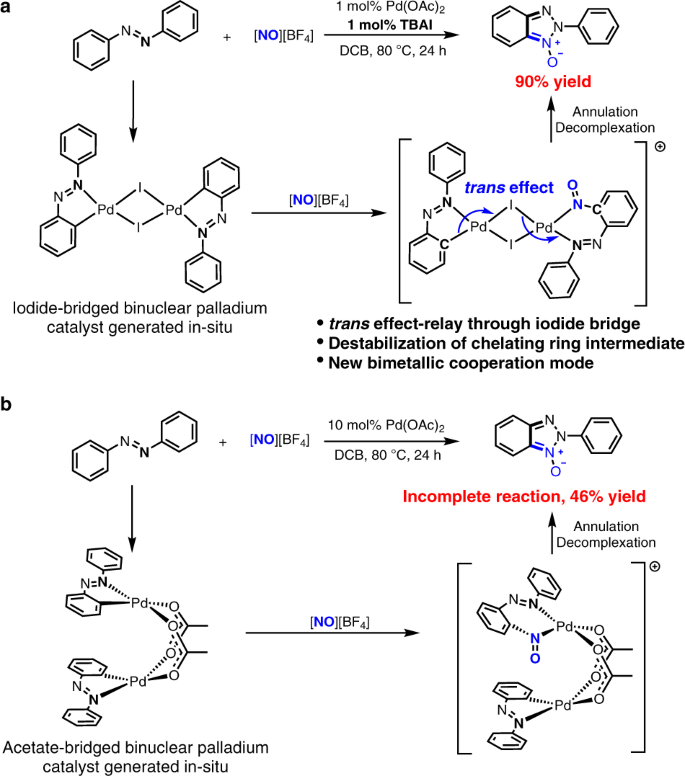
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex | Communications Chemistry
![PDF] Heterogeneous iota carrageenan-based palladium catalysts for organic synthesis | Semantic Scholar PDF] Heterogeneous iota carrageenan-based palladium catalysts for organic synthesis | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b2950915b67892c3c21614a2b444aa27eb3a44a1/7-Table2-1.png)
PDF] Heterogeneous iota carrageenan-based palladium catalysts for organic synthesis | Semantic Scholar
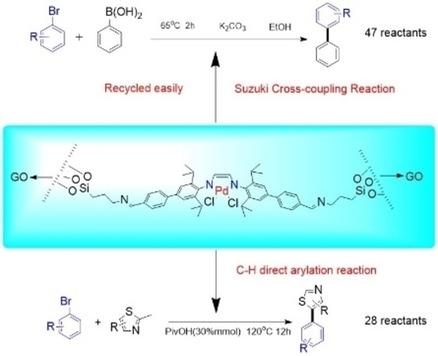
Fabrication and Application of Graphene Supported Diimine‐Palladium Complex Catalyst for Organic Synthesis - ChemistrySelect - X-MOL